___________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: [email protected]

Drug discovery by the medical, biotechnology and pharmaceutical industries is the lengthy, expensive and complex process whereby possible new drugs and medications are detected, primarily to treat diseases and conditions with a currently unmet clinical need.
Throughout history, new therapies to treat disease and prevent illness were discovered through the identification of an 'active' ingredient from traditional remedies, biological sources such as plants, or simply by chance. This active substance was then used to identify a target in the body. Examples include morphine, the active ingredient in opium from poppies, and digoxin which comes from the foxglove plant.
Later, the process reversed and small molecules were created to target a specific pathway or process in the body. This led to a much higher success rate and created drugs that are still used today such as beta-blockers, antibiotics and asthma treatments.

The process of finding a new drug against a chosen target for a particular disease usually involves something called high-throughput screening (HTS). This is a process where large libraries of chemicals are tested for their ability to modify the target; in the case of receptors, their ability to inhibit or stimulate, or by switching on or off a process such as protein kinase activity. This type of screening also reveals how selective the compounds are for the chosen target. The ideal situation is to find a molecule which will interfere with only the chosen target, but not other, related targets. The more unrelated targets a compound affects the more likely it is to have toxic effects.
Drugs and medicines fail to make it to market mainly for two reasons; the first is that they prove to be ineffective, and the second is that they are unsafe for patients. Therefore, modern drug discovery involves an extensive process which includes identifying screening hits, medicinal chemistry and optimisation of those hits to increase the affinity, selectivity (to reduce the potential of side effects), efficacy/potency, metabolic stability (to increase the half-life), and oral bioavailability. Once a compound that fulfills all of these requirements has been identified, it will begin the process of drug development prior to clinical trials.
Modern drug discovery is thus usually a very expensive process that involves large investments by industry corporations as well as national governments (who provide grants and loan guarantees). And despite advances in technology and understanding of biological systems, drug discovery is still a lengthy, expensive and complicated process with low rate of new therapeutic discovery.
Reducing the time and cost applied to any one of the processes involved in drug discovery is a clear goal of anyone involved in the drug discovery field and this is where Diamond can play a key role. Advances in techniques, automation, data collection and processing, and technology have all led to a reduction in the time taken to prepare, screen and collect data on a sample, allowing more potential targets to be assessed and increasing the chances of getting a 'hit'. Techniques such as macromolecular crystallography (MX), fragment screening and electron microscopy all now play key roles in the drug discovery process so let us take you by the hand and lead you through each technique.
Crystallography is the experimental science of determining the arrangement of atoms in a crystalline solid, which can be found everywhere in nature; from salt to snowflakes to gemstones. In 1912, Max von Laue and colleagues at the University of Munich, Germany came up with the idea to send a beam of X-rays through a copper sulphate crystal and record the results on photographic plates. The results showed diffraction spots surrounding the central spot of the primary beam.
Within a year of this discovery, in 1912, William Henry Bragg and son Lawrence had exploited the phenomenon to solve the first crystal structure and create a mathematical formula, Bragg’s Law, which showed how to work out the atomic structure of a sample based on the diffraction pattern it produced when exposed to X-rays. The discoveries of von Laue and Bragg gave birth to two new sciences, X-ray crystallography and X-ray spectroscopy, and resulted in two Nobel Prizes. Crystallography has to date, directly or indirectly, produced the largest number of Nobel Laureates throughout history, up to now 29 Prizes for 48 Laureates..
The Braggs also mentored a dream team of crystallographers who went on to work out the structures of a huge range of molecules. From Kathleen Lonsdale, JD Bernal, Dorothy Hodgkin, to David Phillips, John Kendrew, and Max Perutz. Also, Rosalind Franklin and others even helped map the structure of DNA, probably the most famous result of X-ray crystallography.
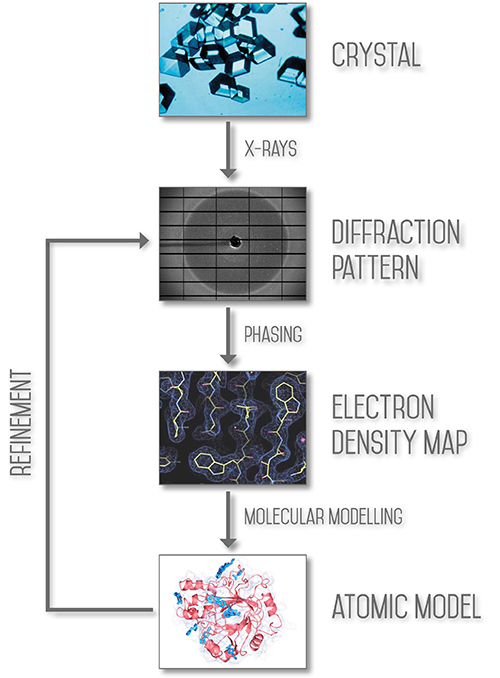
Macromolecular Crystallography (MX, also referred to as Protein Crystallography or PX) is the most powerful method for determining the atomic three dimensional structures of large biological molecules. It is a vital tool for linking structure with function, for rational drug design, for investigating protein folding and for relating other structural information, such as evolutionary relationships, from biological molecules.
The aim of X-ray crystallography is to obtain a three dimensional molecular structure from a crystal. A purified sample at high concentration is crystallised and the crystals are exposed to an X-ray beam, usually under very cold, cryogenic conditions to protect the sample from radiation damage. The resulting diffraction patterns can then be processed, initially to yield information about the crystal packing symmetry and the size of the repeating unit that forms the crystal. This is obtained from the pattern of the diffraction spots.
The intensities of the spots can be used to determine the “structure factors” from which a map of the electron density can be calculated. Various methods can be used to improve the quality of this map until it is clear enough to build a molecular structure (using the protein sequence in the case of macromolecular crystallography).
The MX beamlines here at Diamond allow structure determination by molecular replacement, where the structure of a related molecule is already known, by standard isomorphism replacement with heavy atoms and by Single wavelength Anomalous Dispersion (SAD) or Multiwavelength Anomalous Dispersion (MAD) measurements, in which the wavelength of the X-rays is tuned to be near or at the natural absorption wavelength of selected atoms in the macromolecule. This enables the detection of small differences in the diffraction pattern for reflections that are related as Friedel pairs and these differences allow phase determination to determine the structure through crystallographic methods.
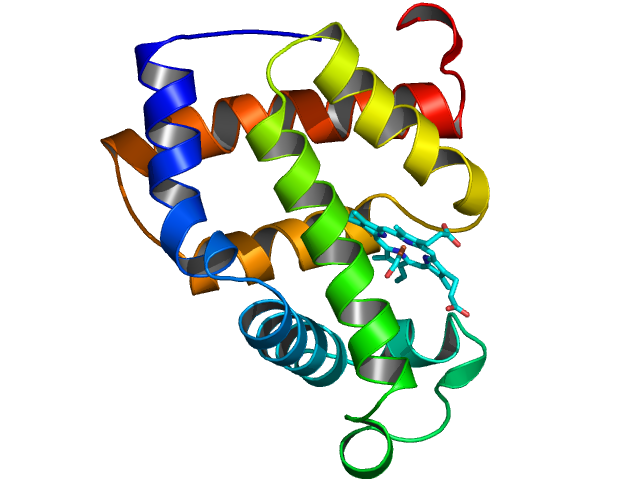 Macromolecules tend to form small, imperfect and weakly diffracting crystals. The high brightness of the X-rays generated at Diamond makes the collection of precise measurements possible. In addition, the state of the art MX beamlines are fitted with advanced robotic systems and software for automated sample handling, crystal centring and data collection along with rapid data processing.
Macromolecules tend to form small, imperfect and weakly diffracting crystals. The high brightness of the X-rays generated at Diamond makes the collection of precise measurements possible. In addition, the state of the art MX beamlines are fitted with advanced robotic systems and software for automated sample handling, crystal centring and data collection along with rapid data processing.
Applications of MX include rational drug design, enzyme mechanisms, supramolecular structure, molecular recognition, nucleic acids, structural genomics and high throughput crystallography. Crystallography experiments at Diamond have recently yielded a potential vaccine for foot-and-mouth disease and have helped scientists to study T-cells: a natural defence mechanism in the body which could provide an effective treatment against cancer.

In fragment screening, scientists test a few hundred tiny chemicals, what they call ‘fragment compounds’, to see if any of them bind to a target and therefore becoming the foundation for what could eventually become a new drug, a 'hit'.
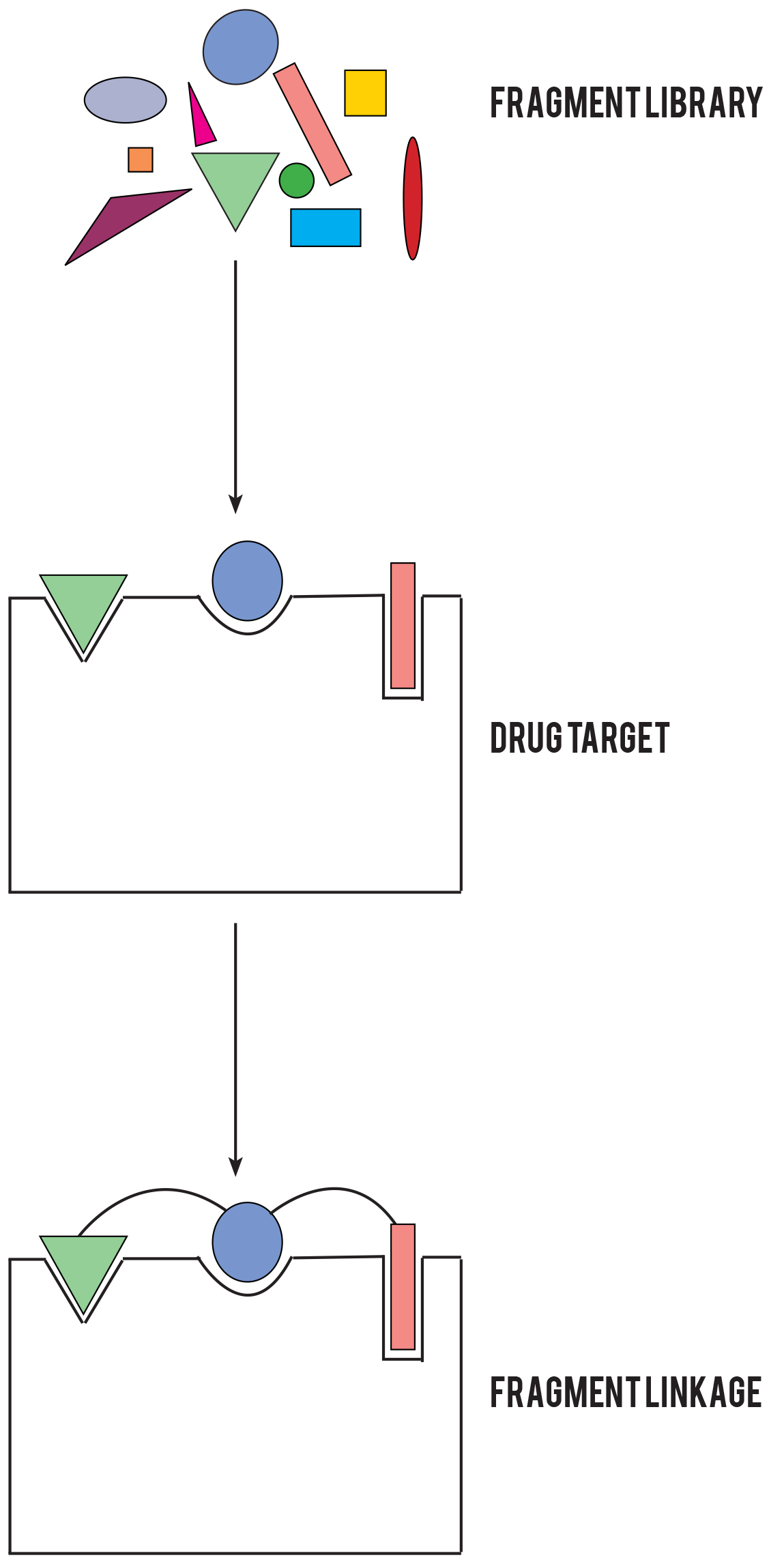
 Diamond Light Source, in collaboration with the Structure Genomics Consortium, has developed a streamlined X-ray fragment screening platform. By seamlessly integrating each step of the fragment screening process, Diamond has tremendously accelerated the X-ray fragment screening approach, while reducing the experimental overheads. Hundreds of compounds are now routinely screened in a matter of days, with a rapid turnover between projects. Users generate the crystals in their home lab, and then come to Diamond to perform soaking and harvesting in I04-1’s Lab 36 (pictured) with Diamond scientists on hand for support. The automated X-ray data collection means that once the crystals have been harvested, users do not need to stay on the beamline and can monitor their experiments remotely.
Diamond Light Source, in collaboration with the Structure Genomics Consortium, has developed a streamlined X-ray fragment screening platform. By seamlessly integrating each step of the fragment screening process, Diamond has tremendously accelerated the X-ray fragment screening approach, while reducing the experimental overheads. Hundreds of compounds are now routinely screened in a matter of days, with a rapid turnover between projects. Users generate the crystals in their home lab, and then come to Diamond to perform soaking and harvesting in I04-1’s Lab 36 (pictured) with Diamond scientists on hand for support. The automated X-ray data collection means that once the crystals have been harvested, users do not need to stay on the beamline and can monitor their experiments remotely.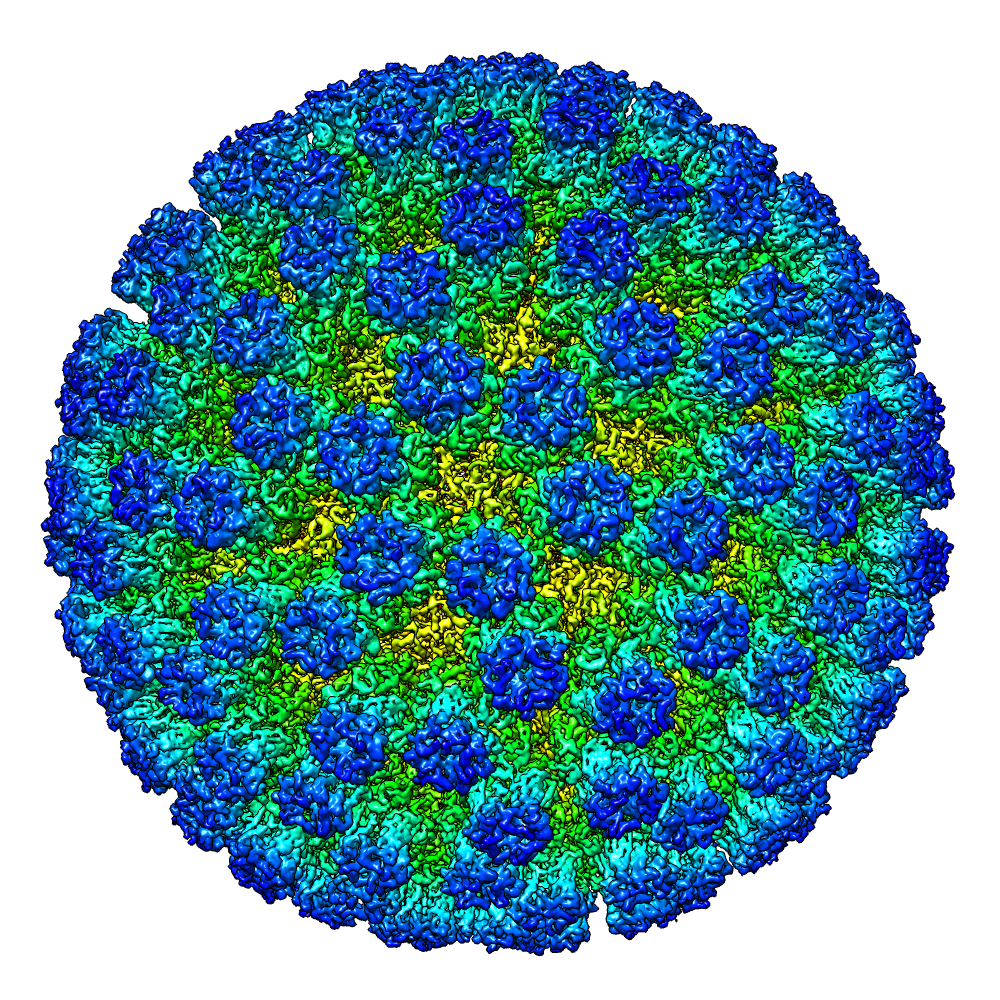 Photons are the basic particles of light and everything we see around us is the result of these particles reflecting from objects and into our eyes. However, some objects are smaller than the wavelength of photons, so they don’t really interact and our eyes just can’t see them. This is where electrons play a part. Because their wavelength is so minute, electrons can interact with tiny objects. So by using electron rather than light microscopes, we can delve even deeper into cellular and molecular structures – about 5,000 times deeper to be precise.
Photons are the basic particles of light and everything we see around us is the result of these particles reflecting from objects and into our eyes. However, some objects are smaller than the wavelength of photons, so they don’t really interact and our eyes just can’t see them. This is where electrons play a part. Because their wavelength is so minute, electrons can interact with tiny objects. So by using electron rather than light microscopes, we can delve even deeper into cellular and molecular structures – about 5,000 times deeper to be precise.
So how did this discovery come about. Well, it was the development of the cathode ray tube that fundamentally changed the way that researchers looked at things; by using electrons instead of light. In 1892, Heinrich Hertz (yes him from the unit of frequency) demonstrated that cathode rays could penetrate very thin metal foil such as aluminium, suggesting that cathode rays were a form of wave motion. But it was not until 1926 that Hans Busch, another German physicist, showed theoretically that a short solenoid (a type of electromagnet) converges a beam of electrons in the same way that glass can converge rays from the sun, that a direct comparison was made between light and electron beams.
Today there are a number different varieties of electron microscopy, but here at Diamond we specialise in Cryo-electron microscopy. Cryo-electron microscopy is a method for looking at frozen-hydrated specimens at cryogenic temperatures (around -200°C). A tiny drop of the sample is placed onto a copper grid, the sample is 'blotted' to leave a thin layer of molecules and then plunged into something like liquid ethane to freeze it so fast that ice crystals can't form. The grid containing the the frozen molecules is then loaded into the electron microscope and the molecules, when shot with the electron beam, leave a unique "shadow" on the detector. These "shadows" contain all the 3-dimensional information of the molecule, compressed into a 2D image. By combining all the images of molecules in all their various orientations, a 3D structure can be created.
 Regular electron microscopy requires samples to be prepared in complex ways – techniques include coating samples in substances that protect them from radiation, sectioning them into tiny slices, or dehydrating them to prevent the interaction between electrons and water molecules. But with cryo-EM, samples don’t require this sort of preparation – they can simply be frozen and then studied in their normal state. This means that scientists can see biological elements as a whole and in an active state. Furthermore, rather than studying individual components of a sample and piecing a wider picture together, cryo-EM enables scientists to look at big, complex biological systems.
Regular electron microscopy requires samples to be prepared in complex ways – techniques include coating samples in substances that protect them from radiation, sectioning them into tiny slices, or dehydrating them to prevent the interaction between electrons and water molecules. But with cryo-EM, samples don’t require this sort of preparation – they can simply be frozen and then studied in their normal state. This means that scientists can see biological elements as a whole and in an active state. Furthermore, rather than studying individual components of a sample and piecing a wider picture together, cryo-EM enables scientists to look at big, complex biological systems.
 Cryo-EM isn’t perfect. It produces lower resolution images than some other techniques, such as X-ray crystallography which we've already discussed. But the real strength of cryo-EM lies in its versatility. The technique allows scientists to study objects that – because of their size, complexity, or sheer awkwardness – would be virtually impossible to scrutinise with other techniques. It’s quick and flexible; and when used in tandem with other techniques, cryo-EM is a supremely powerful tool.
Cryo-EM isn’t perfect. It produces lower resolution images than some other techniques, such as X-ray crystallography which we've already discussed. But the real strength of cryo-EM lies in its versatility. The technique allows scientists to study objects that – because of their size, complexity, or sheer awkwardness – would be virtually impossible to scrutinise with other techniques. It’s quick and flexible; and when used in tandem with other techniques, cryo-EM is a supremely powerful tool.
Here at Diamond we can offer our industrial clients onsite access to eBIC (electron Bio-Imaging Centre). Powerful cryo-electron microscopes allow users to investigate the structure of individual cells and to visualise single bio-molecules, exploiting techniques that are rarely available at home laboratories. Along with state-of-the-art facilities, the centre provides training courses for structural and cell biologists, and remains cutting edge though strong, in-house research.
Below are some examples of how industrial and academic scientist have applied these drug discovery techniques to their research. There are loads more on our Case Study Pages.
We’re always happy to discuss any enquiries or talk about ways in which access to Diamond’s facilities may be beneficial to your business so please do give us a call on 01235 778797 or send us an e-mail. You can keep in touch with the latest development by following us on Twitter @DiamondILO orLinkedIn.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.