___________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: [email protected]
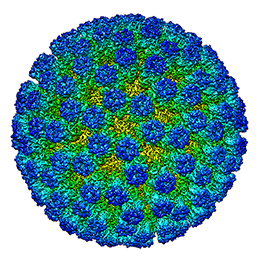 and assemblies. It is a very effective method for studying non-crystalline, dynamic or large macromolecules in their near native state as the often difficult step of crystallisation is avoided. It is therefore of much interest to the pharmaceutical sector.
and assemblies. It is a very effective method for studying non-crystalline, dynamic or large macromolecules in their near native state as the often difficult step of crystallisation is avoided. It is therefore of much interest to the pharmaceutical sector. 1931 - The first transmission electron microscope was developed by Max Knoll and Ernst Ruska. This led to the launch, two years later, of an electron microscope that exceeded the resolution attainable with an optical (light) microscope.
1975 – Joachim Frank began work on the algorithms that would enable scientists to analyse 2D images and reconstruct them into sharp 3D structures.
1981 - Jacques Dubochet and Alasdair McDowall introduced the rapid cryo-cooling of individual molecules in a thin layer of vitrified water. This method prevents ice crystallisation, allows biomolecules to retain their shape in a vacuum and protects them from damage caused by the electron beam and dehydration.
1980s - Joachim Frank and Marin van Heel pioneered the use of computational image processing techniques to bring clarity to noisy cryo-EM images, made even more effective with the introduction of the first tools to reconstruct 3D structures.
1990 – In a world’s first, Richard Henderson used an electron microscope to generate a 3D image of a protein at atomic resolution.
2017 - Jacques Dubochet, Joachim Frank and Richard Henderson won the Nobel prize for developing cryo-electron microscopy for the high-resolution structure determination of biomolecules in solution.



Nobel Prize winners – from left:
Richard Henderson1, Jacques Dubochet2, Joachim Frank3
 of low-atomic mass material and being differentially scattered by the charged electrons in the molecules. The electron microscope converts these phase-shifts in the electron wave into contrast visible in the projection images recorded on a detector.
of low-atomic mass material and being differentially scattered by the charged electrons in the molecules. The electron microscope converts these phase-shifts in the electron wave into contrast visible in the projection images recorded on a detector.
While macromolecular crystallography (MX) has traditionally been the method of choice for structure determination to aid drug discovery, with recent advancements in technology -including the development of direct electron detectors and more effective computational image analysis techniques - the usage of cryo-EM has increased significantly.
Cryo-EM is especially effective in the early stages of drug discovery- target identification, validation and characterisation. The technique has also proved invaluable when determining the structure of large and/or dynamic macromolecular assemblies or when studying membrane proteins that are not amenable to crystallisation. It enables samples to be studied in their native state, giving a much richer understanding of structures and their dynamic states; by using this method, different conformational states can be resolved in one experiment.
| Macromolecular Crystallography (MX) | Cryo-electron microscopy |
|---|---|
| The ability to rapidly and reliably provide high-resolution structural data combined with synchrotron and robotics developments means that crystallography provides very high throughput. | The number of samples that can be resolved is considerably lower than with MX, although automation has significantly shortened the time needed for data integration, structure solution and refinement of protein-ligand complexes. |
| MX relies on biological molecules forming ordered structures and battles to study large flexible assemblies. | Cryo-EM enables you to determine the structure of large and/or dynamic macromolecules (including membrane proteins). |
| Obtaining suitable crystals remains a key challenge that can be very time consuming. | Cryo-EM requires much smaller amounts of protein than MX. It can also be used to study demanding proteins, including membrane, heavily glycosylated or flexible proteins/protein complexes. |
| Crystallisation requires high concentrations of solutes and polymers to induce very tight packing of molecules into a crystalline lattice. | Studying molecules in their native state enables scientists to see how dynamic molecules behave in their natural environments. |
| Phase information is lost and has to be reconstructed by experimental phasing, which depends on the accuracy of experimental data collection. | Phase information is directly and experimentally accessible in the reconstructed structures. |
| Structural resolutions typically much higher, achieving up to ~1 Å. | Most reported cryo-EM structures have been at a resolution of 3 Å or lower. |
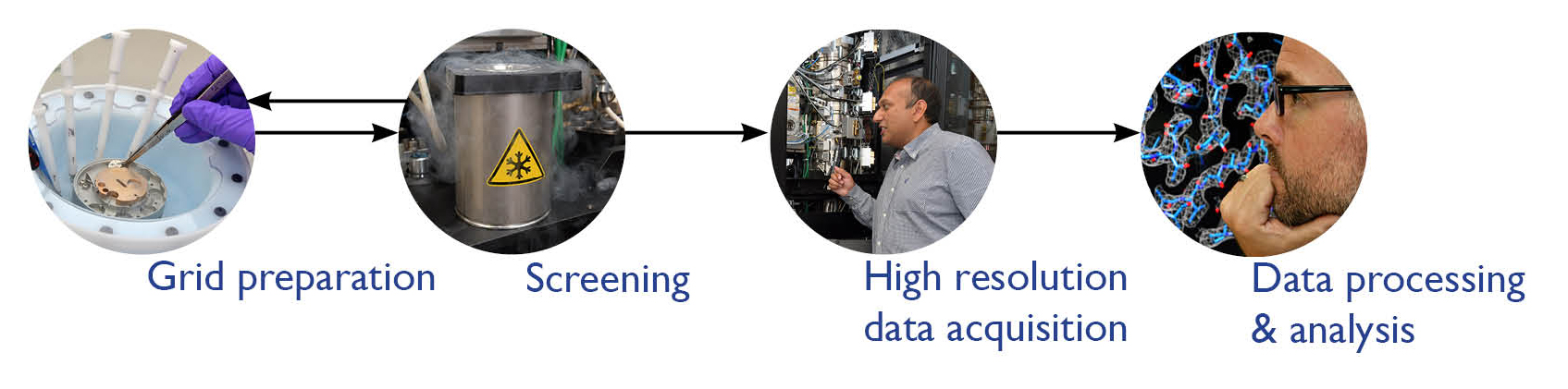
Would you like to know more about cryo-EM and how you can apply it to your research? Do you perhaps have a structural problem that you are unable to solve in your lab? Then please get in touch with the eBIC for Industry team at Diamond.
The eBIC for Industry team at Diamond is a group of professional, experienced in the application of cryo-EM and dedicated to helping scientists and researchers from the biopharma industry access the facilities at eBIC for Industry. We offer a range of services to both experienced groups and new entrants to the field from grid preparation to analysed data.
We’re always happy to discuss any enquiries or talk about ways in which access eBIC for Industry's facilities may be beneficial to your business so please do give us a call on 01235 778797 or send us an e-mail. You can keep in touch with the latest development by following us on Twitter @DiamondILO or LinkedIn
1. Cryo-EM in drug discovery: achievements, limitations and prospects
Jean-Paul Renaud, Ashwin Chari, Claudio Ciferri, Wen-ti Liu, Hervé-William Rémigy, Holger Stark & Christian Wiesmann
doi: 10.1038/nrd2018.77
Nature Reviews Drug Discovery volume 17, pages 471–492 (2018) |
2. How cryo-EM is revolutionizing structural biology
Xiao-chen Bai, Greg McMullan, Sjors H.W. Scheres
https://doi.org/10.1016/j.tibs.2014.10.005
3. Cryo electron microscopy to determine the structure of macromolecular complexes
Marta Carroni and Helen R. Saibil
doi: 10.1.016/j.ymeth.2015.11.023
Methods. 2016 Feb 15; 95: 78–85
4. An introduction to sample preparation and imaging by cryo-electron microscopy for structural biology
Rebecca F. Thompson, Matt Walker, C. Alistair Siebert, Stephen P. Muench, Neil A. Ranson
https://doi.org/10.1016/j.ymeth.2016.02.017
1. By Bengt Nyman from Vaxholm, Sweden (Richard Henderson D81_4486) [CC BY 2.0 (https://creativecommons.org/licenses/by/2.0)], via Wikimedia Commons
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.