Stainless steel is one of our most versatile materials. Its hygienic qualities ensure the safety of medical instruments and implants, and its corrosion-resistant properties make it indispensable in industries from construction to food processing. The corrosion resistance arises from the alloy's chromium content, as the chromium forms a passive film on the surface that can self-heal in the presence of oxygen, shielding the bulk of the material from corrosion. However, the stability of the passive film can be affected by hydrogen absorption, leading to microstructure embrittlement that lowers the stress required for cracks to occur and propagate in the metal. A challenge for the hydrogen energy industry is that high-performance metallic materials are highly susceptible to hydrogen embrittlement. One potential candidate for building a safe hydrogen economy infrastructure is super duplex stainless steel (SDSS).
In work recently published in Applied Surface Science, an international team of researchers used in situ surface-sensitive synchrotron X-ray measurements to investigate the early stages of hydrogen-induced degradation of SDSS occurring at the near surface. Their results show that SDSS’s exceptional resistance to hydrogen embrittlement can be explained by the stability of the passive oxide film, and that the semiconducting property of the passive film plays an important role in hydrogen embrittlement. The authors also conclude that profound in situ experimental characterisation and computational calculation are needed to reveal the complex processes behind material degradation.
Green hydrogen can be used as both a feedstock and energy carrier and has the potential to play a crucial role in the future fossil-free energy landscape. However, high-strength metallic materials are highly susceptible to hydrogen embrittlement (hydrogen-induced material degradation), posing a significant challenge for safe hydrogen storage and transport.
Prof Jinshan Pan, from the KTH Royal Institute of Technology in Sweden, said:
Hydrogen embrittlement is a very important issue for many applications. Different metal materials may have this embrittlement problem. It's really a hundred-years-old challenge. In many cases, the metal surface has a passive film, like an oxide, that allows materials to be used in practice, because otherwise the metals themselves are active.
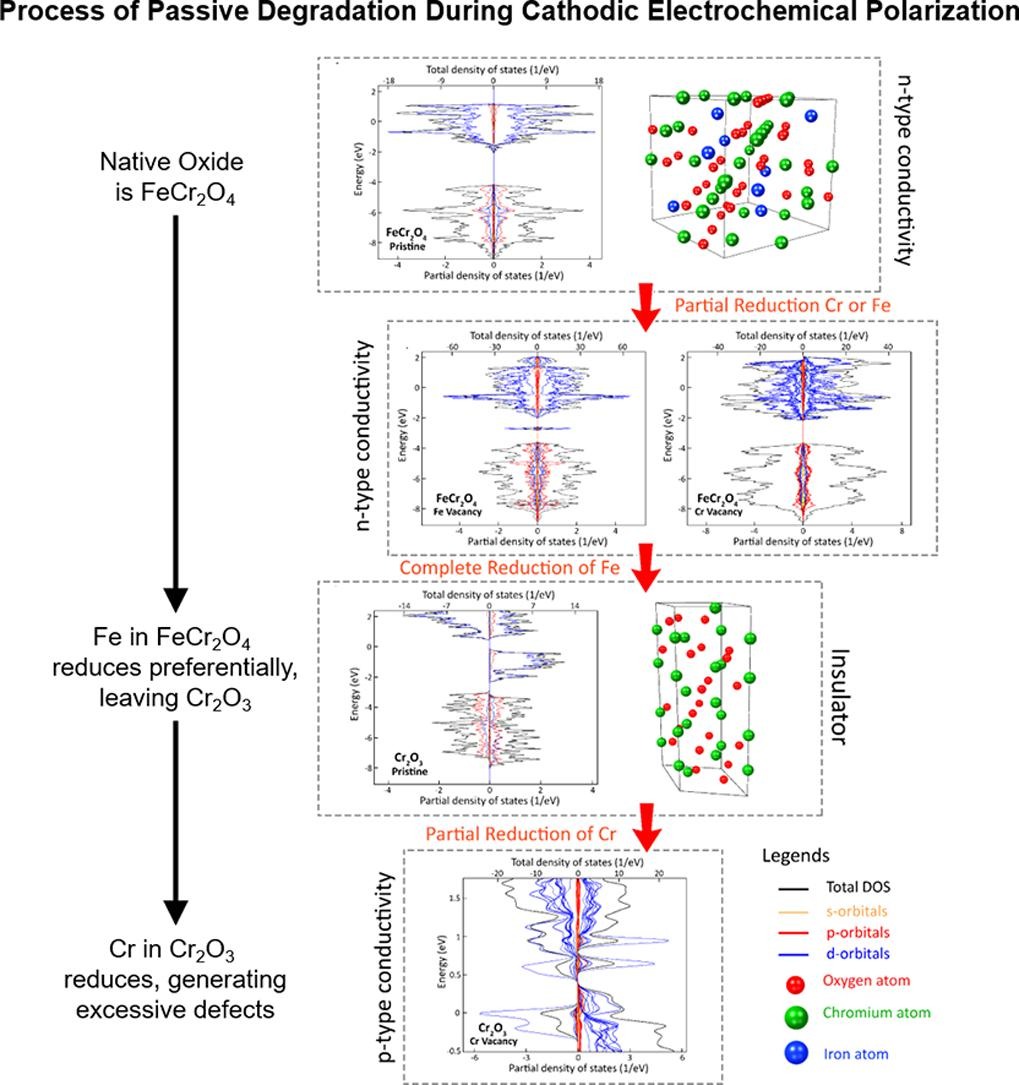
Super duplex stainless steels form a stable passive oxide/hydroxide surface film that is crucial for corrosion resistance, so surface oxide engineering could be an efficient way to combat hydrogen embrittlement. Although the stability of this passive film has been extensively studied under anodic polarisation, its degradation under cathodic polarisation is rarely investigated.
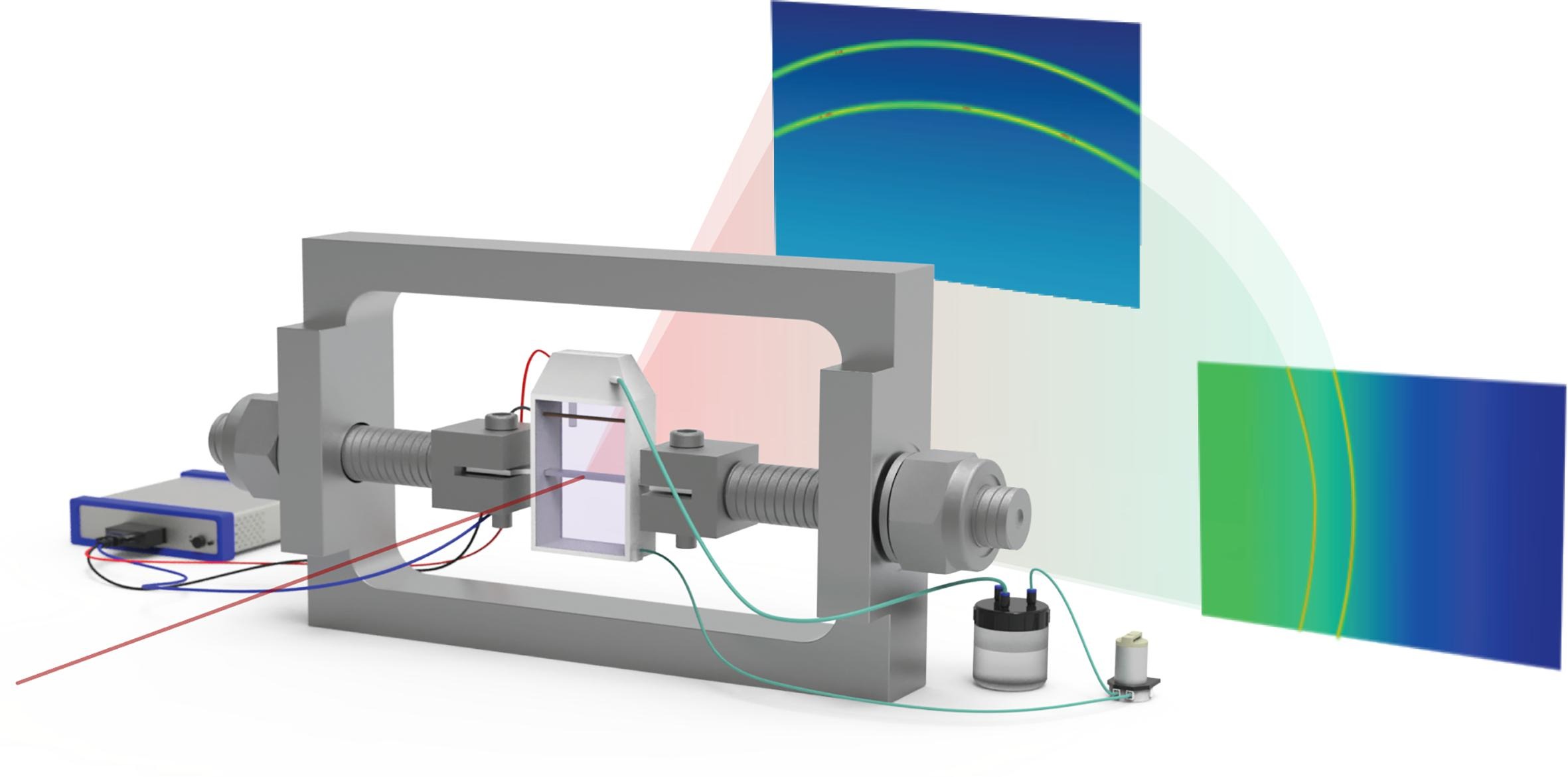
The team’s samples were prepared and kept for two weeks at Diamond ahead of the experiments, exposed to the ambient air to develop an oxide film. On the I07 beamline, they used Gazing-Incidence X-Ray Diffraction (GIXRD) and X-Ray Reflectivity (XRR) measurements to probe the near-surface region to approximately 40 nm and 170 nm during in situ cathodic electrochemical polarisation. Synchrotron-based XRR is a very sensitive technique for measuring the thickness and density of thin surface films. Electrochemical Impedance Spectroscopy (EIS) measurements revealed the electrochemical properties of the surface oxide film.
Prof. Pan continued:
How this surface oxide affects hydrogen embrittlement is poorly known and little studied. It's not easy to study! So we used a very surface sensitive synchrotron technique at Diamond - surface diffraction. And we tried to do that in situ, during hydrogen charging, and even in operando, where we applied mechanical load, and it's like operating conditions. In this way, we could explore how the surface oxides on super duplex stainless steel change under these conditions.
Combined with CALPHAD-based thermochemical modelling and ab initio density functional theory (DFT) calculations, the experimental results show that the absorption of hydrogen causes oxide reduction and ionic defect generation within the passive film, changing its semiconductor properties and causing surface degradation. The surface film weakens and allows atomic hydrogen to infuse into the lattices of the austenite and ferrite phases and cause embrittlement.
Prof. Pan concluded:
This is quite fundamental research. SSDS includes two phases with different microstructures. Manufacturing the highest quality SSDS involves controlling the microstructure in a very balanced way through the whole production process. To do that, you need a deeper knowledge of how to optimise the microstructure, and our research adds to that knowledge. It will also be useful for determining the safe operating conditions for existing materials and provides guidelines for designing new advanced materials.
To find out more about the I07 beamline or discuss potential applications, please contact Principal Beamline Scientist Francesco Carlà: [email protected]
Örnek C et al. Understanding passive film degradation and its effect on hydrogen embrittlement of super duplex stainless steel–synchrotron X-ray and electrochemical measurements combined with CalPhaD and ab-initio computational studies." Applied Surface Science 628 (2023): 157364. DOI: 10.1016/j.apsusc.2023.157364.
Örnek C et al. Metastable precursor structures in hydrogen-infused super duplex stainless steel microstructure–An operando diffraction experiment. Corrosion Science 176 (2020): 109021. DOI:10.1016/j.corsci.2020.109021.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.