Instruments by Science Group
I04-1 Contact
Beamline Phone Number:
+44 (0) 1235 778979
Principal Beamline Scientist:
Frank von Delft
Tel: +44 (0) 1235 778997
E-mail: [email protected]
Science Group Leader
Dave Hall
Email: [email protected]
Tel: +44 (0) 1235 778926
I04-1 Fixed wavelength MX
Status: OperationalDetector: Eiger2 XE 9M
Wavelength: 0.92Å (fixed)
Energy: 13.445 keV
I04-1 is a fixed wavelength monochromatic beamline, with its undulator in I04's straight section. The beamline is aimed at high-throughput data collection for well-diffracting crystals, with off-the-shelf robotics and a stable beam thanks to a simple beamline design. Flux has been significantly increased since the insertion device install in July 2022 offering 3.8x1012 (photons/second) with the 70um apeture.
As of April 2015, the beamline is also offering a world-first, namely routine medium-throughtput fragment screening by crystal structure, including all steps from crystal soaking and harvesting to data analysis. Full details and application process are here.
WEBCAMS
- Live view! (intranet)Goniometer and Experiment Overview
Background
I04-1 delivers a high-throughput approach similair to that pioneered in industry, where it is axiomatic that the best way to study ligand binding involves developing a well-diffracting crystal system, which allows for the rapid X-ray data collection necessary to observe the binding of the many compounds generated by medicinal chemistry. The implication for a beamline is that flexibility is less important than stability and speed. I04-1 is heavily utilised by the XChem platform, as well as being fully availiable to the MX user community for rotational cryogenic experiments.
Research program
The research focus is on taking high-throughput approach to its logical extreme, namely all aspects of implementing and exploring routine medium- throughtput fragment screening by crystal structure. This involves a lot more than just the X-ray experiment, and since late 2012 the beamline has been partnered with the Protein Crystallography group, now at the Centre for Medicince Discovery in University of Oxford.
Developments include:
- Academic and Industrial access to high throughput crystallogrpahic screening
- Rapid synthesis of compounds using automated robotics platforms
- Fragalysis platform - Quick review of Crystallographic fragment data for advancing medicinal chemistry of screened targets
- Development of algorithms for hit identifentifaction in crystallographic screens: Pan-Dataset Density Analysis (PanDDA)
- Development of room temperature crystallographic fragment screening
I04-1 Specification
| Energy | fixed, monochromatic: 0.920Å / 13.53 keV |
| Experimental phasing | SAD: optimal for Br, good for Se |
| Flux (ph/sec) |
3.8 x 1012 (70um aperture, 300mA ring current) 2.5 x 1012 (50um aperture, 300mA ring current) 1.3 x 1012 (30um aperture, 300mA ring current) 9.0 x 1011 (20um aperture, 300mA ring current) 3.0 x 1011 (10um aperture, 300mA ring current) |
|
Beam size options (µm): |
|
| Detector | Eiger 2 XE 9M |
| Maximum resolution (Å) | 1.6 |
| Dataset time, typical | 7.2s (3600 Images, 0.1 degrees, 100% Transmission, 0.002s) |
| Sample changer | Diamond BART with unipucks |
| Sample exchange time | < 20s |
| No. of pins in dewar | 592 |
| Pins and Pucks | SPINE standard pins (only!) and Unipucks |
|
Samples rate (xtals/hour) |
15-30 (manual, user centring) 29 (queued, automatic X-ray centring) |
Please discuss your requirements with a member of the beamline team before your experiment.
SAD phasing at I04-1
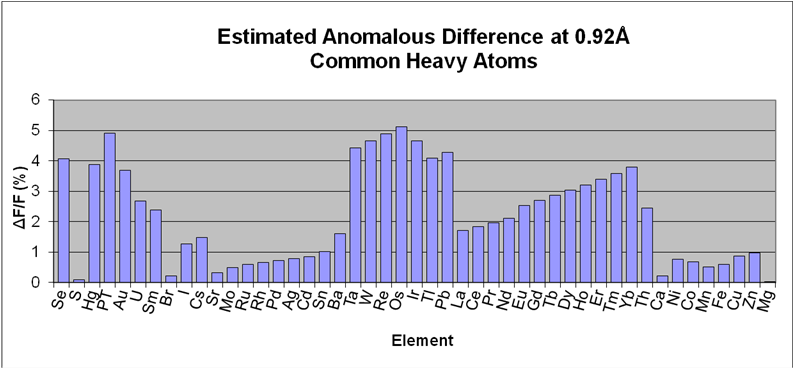
I04-1's wavelength is fixed at the Bromine K edge (E = 13530 eV, λ = 0.9163 Å), where a strong anomalous signal can be measured for most commonly-used heavy atom derivatives (see figure). SAD is usually very effective here - unsurprisingly: thanks to modern detectors and phasing algorithms, tunability is essential only for special cases, especially if the experiment is done carefully (see e.g. Krojer, Pike, von Delft, Acta D 2013)
Estimated anomalous difference at 0.92Å resolution for common elements. These were calculated assuming: for all elements, 1 fully occupied heavy atom site per 300 residues; for Se, 1:42 heavy atoms:residue; and using the formula derived from Smith, J., Curr. Opin. Struct. Biol. vol1, p1002 (1991).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.
.png)